THERAPEUTIC AREAS
KEOSYS PROVIDES SERVICES AND SOLUTIONS IN A VARIETY OF THERAPEUTIC AREAS.
As an Imaging CRO, Keosys has been providing services and solutions in phase I-IV clinical trials in a wide variety of therapeutic areas including oncology, cardiovascular, neurology and gastrointestinal studies. We have experience working with all vendors and imaging modalities, including advanced molecular imaging techniques with a unique set of challenges.

ONCOLOGY
Keosys has supported the imaging component of over 100 clinical studies in oncology, including 3 pivotal efficacy studies that resulted in drug approvals in the United States and Europe. The evaluation criteria applied include RECIST 1.1, iRECIST, mRECIST, RANO, PERCIST, Cheson, Lugano, PCWG3, IMWG and others. Keosys currently provides imaging CRO services in lymphoma studies that are among the world’s largest, involving over 1,000 patients and 165 investigator sites
NUCLEAR MEDICINE
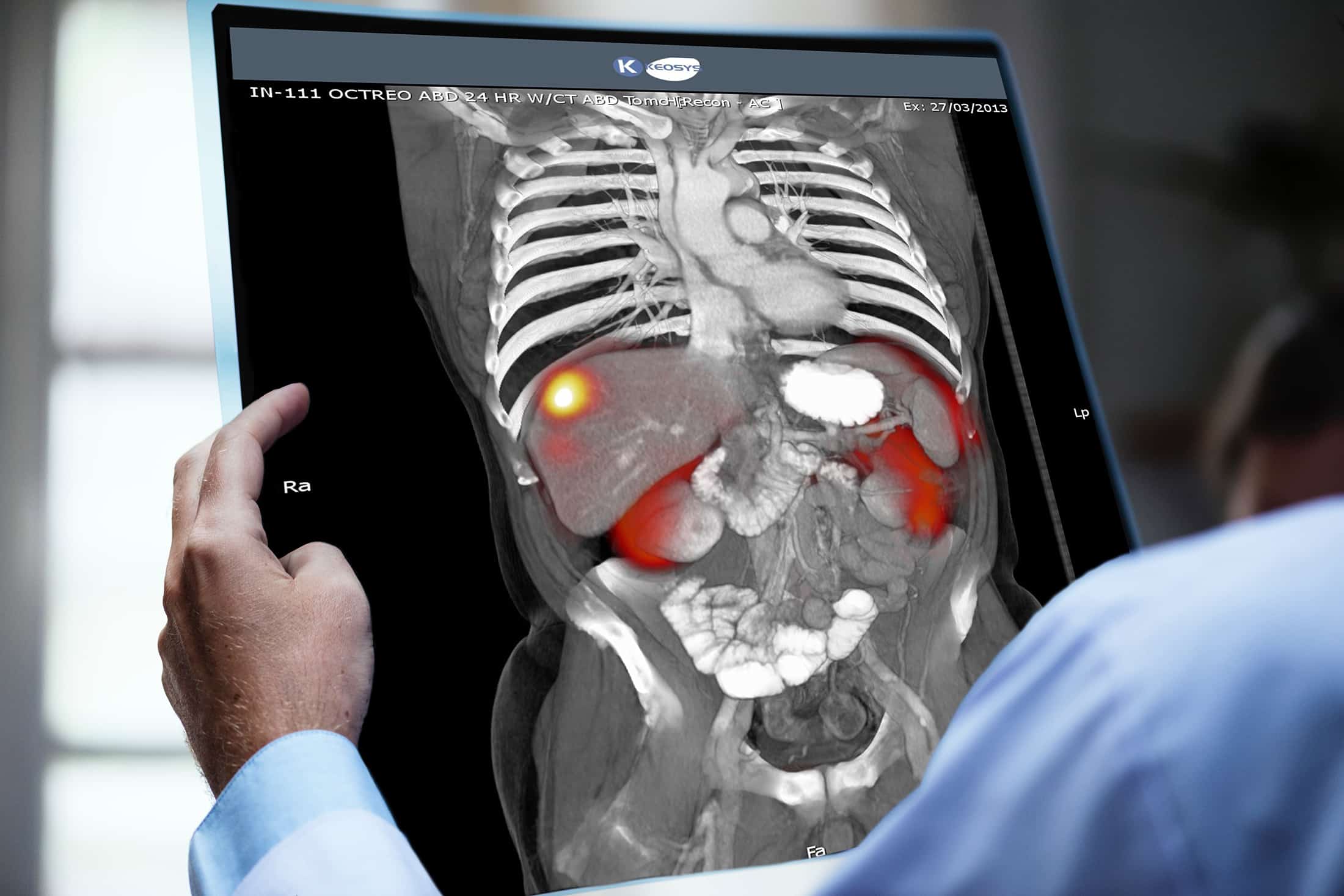
Since its early days, Keosys has been at the forefront of molecular imaging—including the field of nuclear medicine, which uses radioactive materials to diagnose and treat diseases. Our in-house technology and network of radiologists are tailored to the needs of any trial in this domain, including organ dosimetry analysis, peptide radiotracer imaging, and advanced PET/CT imaging.
OTHER AREAS
We have the experience and required tools to work on a wide range of other imaging response criteria in numerous therapeutic areas, including: cardiology, neurology, rheumatology and GI. Our technology and proven methodology can be used for all trials involving DICOM imaging.
MODALITIES
Keosys can help you with trials involving all modalities including CT, MRI, PET, SPECT, CT, X-RAY, colonoscopy, ultrasound and more. Choosing the appropriate modality and imaging endpoint for your trial is a key driver in determining the success of your project. Keosys works closely with the clinical teams during the protocol definition phase in order to make sure all imaging challenges are met.
